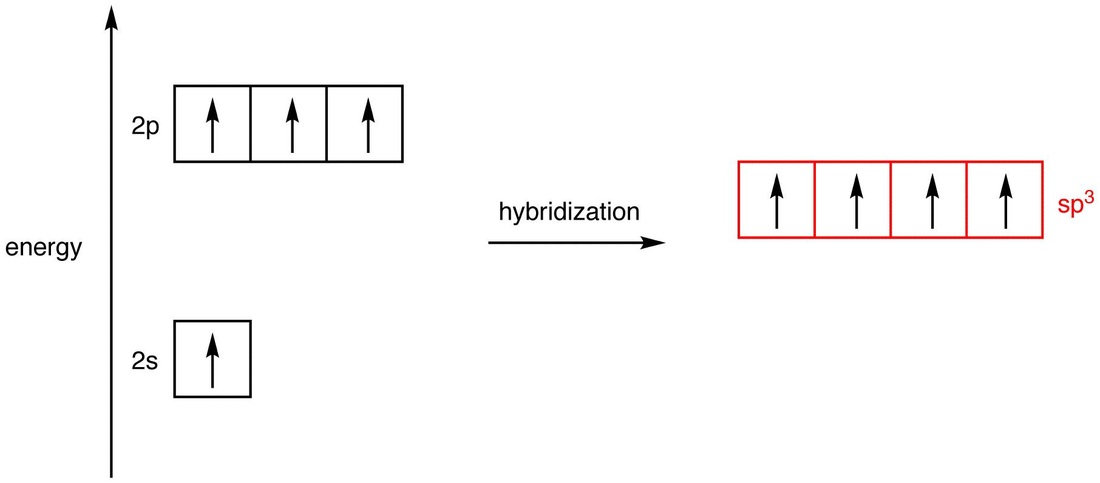
So, the dx^2-dy^2 orbital isused in dsp2 hybridization, along with the s, px and py orbitals to form a square planar geometry.īeside above, what is the difference between sp3d2 and d2sp3? Transition metals may exhibit d2sp3 hybridizationwhere the d orbitals are from the 3d and the s and p orbitals arethe 4s and 3d. The dsp2 hybridization favours a planar geometryin which the four hybridized orbitals lie in the equatorialxy plane, as in ICl4- or XeF4. Hereof, which d orbital is involved in sp3d hybridization and why? In octahedron, the bonds are formed parallelto the x, y, and z-axes, hence dx 2-dy 2 anddz 2 will be used to form the hybrid orbitals. Which d orbitals are involved in sp3d2 andd2sp3 hybridization respectively ? Answer:sp 3 d 2 or d 2sp 3 are hybridisation for theoctahedral geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
